New Therapeutics in Triple-Negative Breast Cancer
Ellie McCluey | April 24, 2023
Triple-negative breast cancer accounts for around 15 percent of all breast cancers. It is distinct from other forms of breast cancer due to the lack of three critical receptors on cancer cells that are common to other forms of breast cancer. The lack of receptors makes it especially aggressive in comparison; it tends to grow and spread quicker and has less variability in treatment options.
Fiona Cherry is a current senior at Vanderbilt majoring in Chemistry who has worked in the Duvall Lab since her freshman year. The Duvall Lab is a biomedical engineering lab that specializes in biomaterials and drug delivery systems. Fiona’s project specifically focuses on finding therapeutics for triple-negative breast cancer. Recent therapeutics for breast cancer have been made to target the three critical receptors that are lacking in triple-negative breast cancer and are thereby ineffective for triple-negative cancer. Fiona’s project is working around these constraints to improve the outlook of patients with this aggressive form of cancer.
Fiona’s project develops polymer-based nanoparticles that can deliver silencing RNA (siRNA) into cancer cells. The nanoparticles are taken up by cancer cells and target upregulated mRNAs and prevent their function, thereby preventing aberrant protein synthesis of key cancer genes. These silencing RNAs cannot be directly administered into the blood because they are degraded quickly and cleared by the kidneys. The nanoparticles created in the Duvall Lab are designed to safely deliver the siRNA to the tumor site and enter the cell through endocytosis.
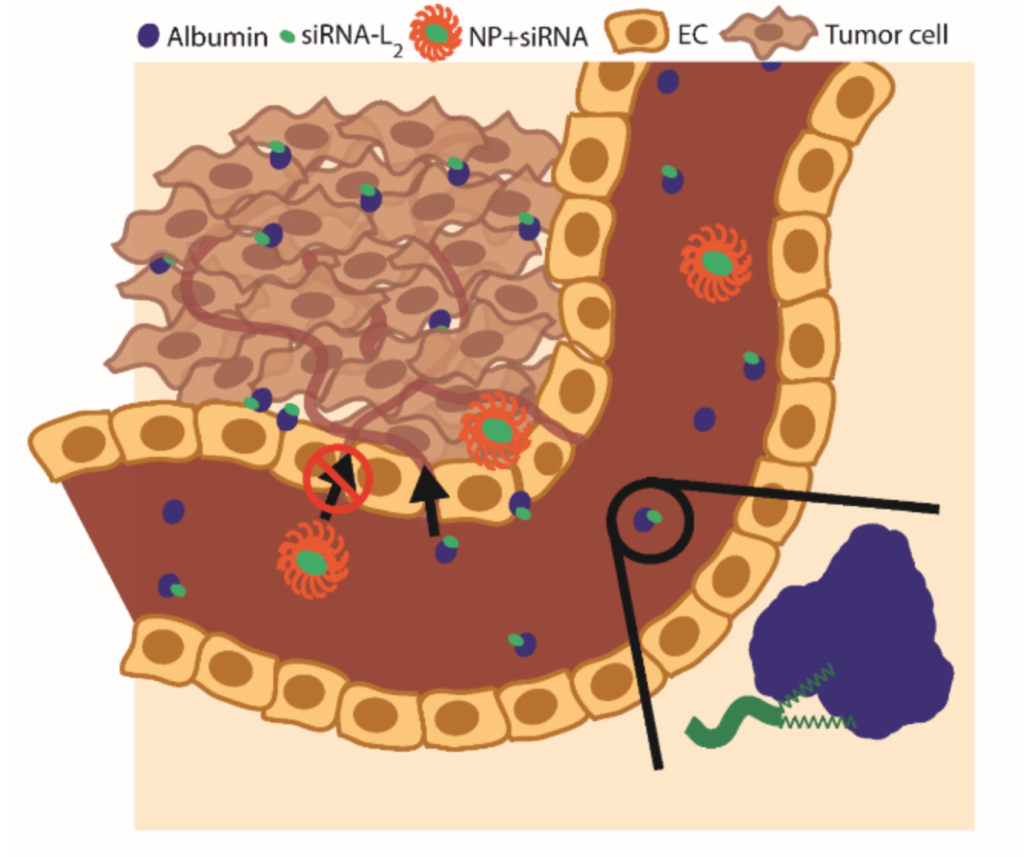
Once inside the cell, the nanoparticles need a way of safely releasing the siRNAs inside. The nanoparticles developed in the lab are pH responsive so thatwithin the endosome, the nanoparticles display disruptive behavior that allows the endosome to burst and release the siRNA into the cytoplasm. From there, the siRNA binds to a specific mRNA that is a precursor for a protein in the PI3/AKT/mTOR pathway, an important regulator of the cell cycle that becomes upregulated in breast cancer. The silencing of a specific protein in this pathway diminishes the cancer’s cellular proliferation. By decreasing the expression of a specific protein in this pathway, the lab hopes to see a decrease in the cancer progression and improve the future outlook of people with triple-negative breast cancer.
When asked to sum up her experience in the Duvall Lab, Fiona stated, “It’s been really cool to stay in one lab [for the past few years] and see people come and go, projects come and go, and be able to track my progress [over time] […] I can look back and see lots of personal and technical growth that I’ve gained through this experience”. The research conducted in the Duvall Lab will hopefully benefit thousands of people with triple-negative cancer by pushing science and medicine to consider new therapeutic approaches to cancer.
Works Cited