Eric Sha | October 28, 2022
Our understanding of how to treat and prevent the COVID-19 virus has grown continuously throughout the pandemic. One of the most innovative methods for treating and preventing COVID-19 that arose during this time is the use of monoclonal antibodies. Developed from research performed at Vanderbilt, monoclonal antibodies are already being used to save lives, and have laid the groundwork for future battles against viral infections.
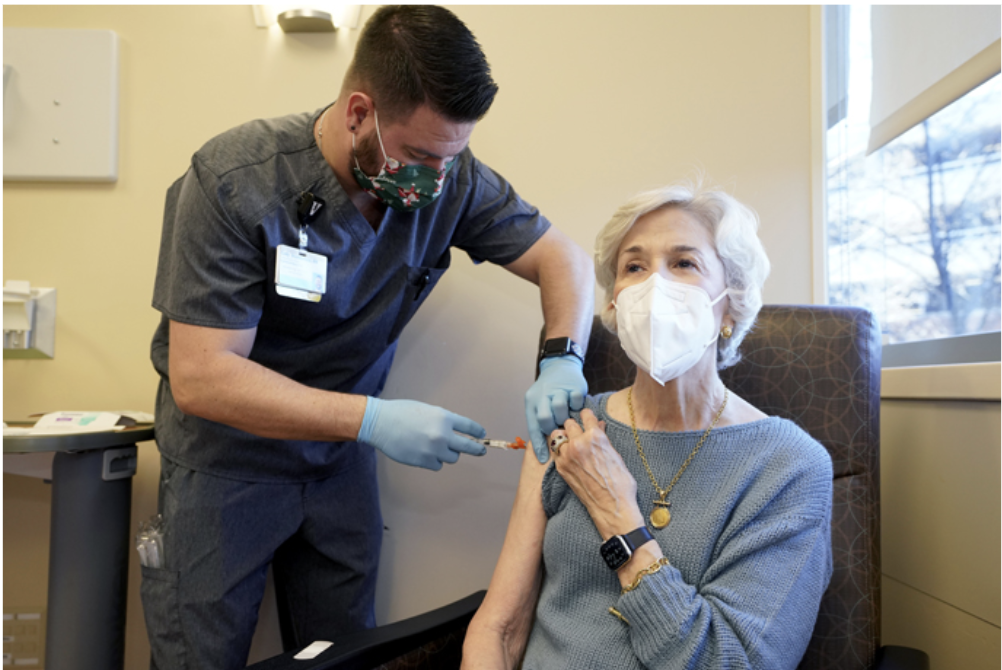
Image to the right: An immunocompromised patient receiving AstraZeneca’s Evusheld-the first set of antibodies grown in a lab to prevent Covid-19
What are monoclonal antibodies?
Monoclonal antibodies work similarly to regular antibodies, which are generated by our body’s immune system. These proteins identify and bind to spike proteins of various viruses, such as SARS-CoV-2. This binding neutralizes the virus and prevents it from entering a cell to replicate itself.
What makes monoclonal antibodies different from regular antibodies? Rather than being naturally produced by the immune system, monoclonal antibodies are made in labs and infused into the patient. They are helpful because people with various autoimmune diseases or immunodeficiencies, such as HIV, may not be able to naturally generate antibodies against SARS-CoV-2, even after receiving a vaccine. Without the necessary antibodies, these people are at a heightened risk of getting very sick or even dying from COVID-19. Therefore, it is paramount that they receive monoclonal antibodies to fight off the infection.
Monoclonal Antibody Research at Vanderbilt
The neutralizing monoclonal antibodies that provided a framework for both monoclonal antibody and vaccine design were first presented in a 2020 Nature paper authored by Drs. James E. Crowe Jr, Robert Carnahan, and others at the Vanderbilt Vaccine Center. In this paper, the researchers presented two monoclonal antibodies (COV2-2196 and COV2-2130) that attached to the spike protein of and neutralized the SARS-CoV-2 virus.
When tested in mouse models infected with SARS-CoV-2, the researchers found that the addition of these antibodies reduced the viral burden and levels of inflammation in the lungs. These results led to the development of the global pharmaceutical company AstraZeneca’s monoclonal antibody treatment for COVID-19, Evusheld. Evusheld was approved by the FDA in 2021 for the use of those with compromised immune systems or a history of severe adverse reactions to a COVID-19 vaccine. In a clinical trial, Evusheld recipients had a 77% reduced risk of developing COVID-19 compared to the control group. For those who are immunocompromised, this represents a potentially life-saving alternative to the COVID-19 vaccine.
Just last month, Evusheld received the gold medal at the 2022 R&D 100 Awards program. The drug was selected for the prize by a panel of scientists, engineers and entrepreneurs from industry, government and academia. Crowe stated, “We’re grateful that the research and development community has recognized the teams at Vanderbilt and AstraZeneca that put forth so much effort under adverse circumstances in 2020 to bring this antibody drug to approval. Fortunately, the antibodies are still bringing benefit to patients at risk of severe infection.” The award represents these benefits, as well as the culmination of years of hard work done in the lab, from discovery to drug design to being approved and used in hospitals.
Works Cited
File:Monoclonal Antibody.Jpg – Wikimedia Commons. https://commons.wikimedia.org/wiki/File:Monoclonal_antibody.jpg. Accessed 10 Oct. 2022.
“Frequently Asked Questions on the Emergency Use Authorization for Evusheld (tixagevimab co-packaged with cilgavimab) for Pre-exposure Prophylaxis (PrEP) of COVID-19” United States Food and Drug Administration https://www.fda.gov/media/154703/download#:~:text=In%20the%20primary%20analysis%2C%20Evusheld,Evusheld%20recipients%20through%20six%20months.
Snyder, Bill. “COVID Antibody Research Conducted at VUMC Lands National Award.” Vanderbilt University, https://news.vumc.org/2022/08/24/covid-antibody-research-conducted-at-vumc-lands-national-award/. Accessed 26 Sept. 2022.
Snyder, Bill. “Patient Receives Antibodies Discovered at Vanderbilt to Prevent COVID-19 Illness.” Vanderbilt University, https://news.vumc.org/2021/12/23/patient-receives-antibodies-discovered-at-vanderbilt-to-prevent-covid-19-illness/. Accessed 10 Oct. 2022.
“What Is Monoclonal Antibody Treatment and How Is It Used for COVID-19?” Healthline, 21 Sept. 2022, https://www.healthline.com/health/what-is-monoclonal-antibody-treatment.
Zost, Seth J., et al. “Potently Neutralizing and Protective Human Antibodies against SARS-CoV-2.” Nature, vol. 584, no. 7821, Aug. 2020, pp. 443–49. www.nature.com, https://doi.org/10.1038/s41586-020-2548-6.